英国UKCA认证

UKCA (UK Conformity Assessed) 英国合规性评估
根据英国政府颁布的公告告知,从2021年1月1日开始,UKCA标志将正式启动使用在产品上。未来大多数在CE管控范围内的产品进入英国都需要补充UKCA标志。UKCA是一种在英国脱欧后新的英国产品标志,用于在英国(英格兰,威尔士和苏格兰)投放市场的商品。它涵盖了以前需要CE标志的大多数商品,称为“新方法”商品。
2019年到2020年脱欧的过渡期期间,英国政府已向英国制造商发布了有关将产品投放到英国市场以及将产品投放到欧盟市场(如果英国没有达成协议就离开欧盟)的指南。政府已经针对医疗器械和建筑产品发布了类似的建议。
According to the announcement issued by the British government, from January 1, 2021, the UKCA logo will be officially used on products. In the future, most products within the scope of CE control will need to supplement the UKCA mark when entering the UK. is a new British product mark for goods placed on the market in the United Kingdom (England, Wales and Scotland). It covers most goods that previously required CE marking, known as "New Approach" goods.
During the transition period from 2019 to 2020, the UK government has issued guidance to UK manufacturers on putting their products on the UK market and on the EU market if the UK leaves the EU without a deal.
The government has issued similar recommendations for medical devices and construction products.

1. UKCA和CE到底有什么分别呢?
What are the differences between CE marking and UKCA marking?
回答前我们先要知道英国脱欧基础,什么是UKCA?
First of all, we need to know the basis of Brexit and what is UKCA?
UKCA(英国合规性评估)标志将是新的英国产品标识,如果英国在没有脱欧协议的情况下退出欧盟,仍然可以在投放英国市场的产品上使用 CE 标志。
If the UK leaves the EU without a deal in most cases you will still be able to use the CE marking for products being placed on the UK market.
UKCA它不会在欧盟市场上得到认可。需要CE标志的产品仍需要CE标志才能在欧盟销售。
It will not be recognised in the EU market.Products that need CE mark still need CE mark before they can be sold in the EU.
在英国退欧后,欧盟的主要产品标准将被全新的英国等同物(UKCA标志)取代,过渡期间,旅行和贸易将继续进行,而不会产生额外费用或支票。但是,在过渡期结束时,英国将退出欧盟市场和关税同盟。新的英国合格评定(UKCA)法规有望取代欧洲CE认证框架。用于英国市场上出售商品的新产品标记是UKCA标记。
After Brexit, the EU's main product standards will be replaced by a new UK equivalent (UKCA logo), and travel and trade will continue during the transition without additional fees or cheques. However, at the end of the transition period, the UK will leave the EU market and customs union. The new UK Conformity Assessment (UKCA) legislation is expected to replace the European CE certification framework. The new product label used for goods sold in the UK market is the UKCA label.
未来英国和欧盟的贸易还会存在,两者会以合作伙伴的状态继续进行贸易交流
In the future, trade between the UK and the EU will still exist, and the two will continue to trade as partners
2020年1月1日启动正式UKCA标志企划后,使用了英国批准的机构(相当于英国的欧盟认证机构),并且只希望将其产品投放到英国市场将需要注册UKCA标志。CE证书在英国将需要更替成UKCA才可进口英国市场
After the launch of the official UKCA logo scheme on 1 January 2020, using a UK approved body (equivalent to the EU certification body in the UK), and only wishing to launch their products in the UK market will require registration of the UKCA logo.CE certificate in the UK will need to be replaced by UKCA before it can be imported into the UK market
2. UKCA注册条款未来会和CE差不多吗?
Will the UKCA registration terms be similar to CE in the future?
是的,UKCA未来还会接受EN标准,总体来看注册流程还是依赖CE体系。
值得注意的是,化学药品,药品,化妆品和食品没有CE认证标志,将来可能也不需要UKCA标志-这些类型的商品受单独的标签要求覆盖。(关注珩渥检测公众号及时接受最新检测认证消息)
Yes, the UKCA will accept the EN standard in the future.
In general, the registration process still depends on the CE system.
It should be noted that chemicals, pharmaceuticals, cosmetics and foods do not have the CE mark and may not need the UKCA mark in the future - these types of goods are covered by separate labelling requirements.
(Pay attention to Headway's official account to accept the latest information of testing certification in a timely manner)
3. UKCA和CE标志之间有一些重要的区别需要注意。
There are some important differences between the UKCA and CE marks to note.
如果发生“无交易”退欧,则修订英国产品合规性法规的法定文书的副本可在此处找到。并且在英国市场上投放的产品仍然可以使用CE标志。
上述建议仅在英国无协议离开欧盟的情况下适用。任何提款协议或未来关系的规定均受这些协议约束。还是需要观看未来政府决定
使用英国认证机构的制造商应尽快与该机构联系,以了解其“英国退欧后”计划,或者是否确实更适合使用UKCA标记
通过欧盟供应链进行分销的制造商应了解,该供应链的经济运营商地位将发生变化,这可能需要更改包装。还提醒机械制造商,如果他们打算在“无协议退欧”之后将产品投放到欧盟市场,则应提名负责编制技术文件的人员,该人必须位于欧盟内部。
In the event of a no Deal Brexit, a copy of the statutory instrument amending the UK's product compliance regulations can be found here.
And products on the UK market can still use the CE mark.
The advice would only apply if The UK left the EU without a deal.
The provisions of any withdrawal agreement or future relationship shall be governed by these agreements.
It is still up to the future government to decide
Manufacturers using the UKCA should contact the BODY as soon as possible to find out about their post-Brexit plans or whether they are indeed better suited to using the UKCA mark
Manufacturers distributing through the EU supply chain should be aware that the economic operator status of that supply chain will change, which may require a change in packaging.
Machinery makers have also been warned that if they intend to launch their products on the EU market after a "no agreement Brexit", they should nominate someone responsible for preparing the technical documentation, which must be within the EU.
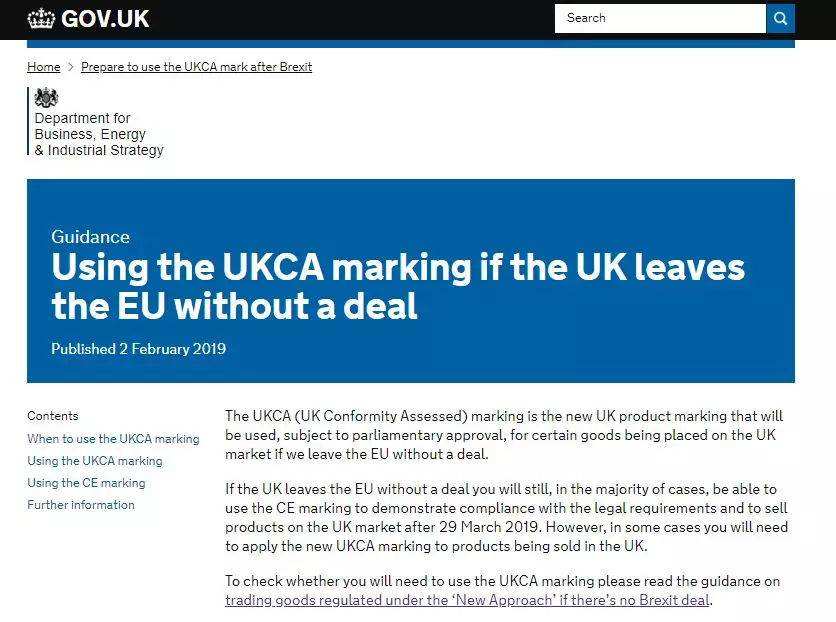

脱欧之后厂家需要注意什么?
What should manufacturers pay attention to after Brexit?
2019年3月29日后,以下产品在英国市场销售将不可以继续使用欧盟规范及符合性标志:
After March 29, 2019, the following products will no longer be allowed to use eu norms and compliance marks in the UK market:
1、 汽车(车辆型式认证);
Automobile (Vehicle type certification);
2、 航空航天(航空安全);
Aerospace (Aviation safety);
3、 医药产品(批量测试药物;药品、医疗器械和临床试验;药品、医疗器械和临床试验的进一步规范;提价医疗产品的监管信息);
Pharmaceutical products (batch test drugs;Drugs, medical devices and clinical trials;Further specification of drugs, medical devices and clinical trials;
4、 医疗器械(药品、医疗器械和临床试验;提交医疗产品的监管信息);
5、 Regulatory information of medical products with price increases); Medical devices (drugs, medical devices and clinical trials; Submit regulatory information on medical products);
6、 化学品(管理化学品、化学品的分类、标签和包装);
Chemicals (chemicals to be managed, classification, labelling and packaging of chemicals);
7、 受国家法规管制的物品(非协调物品)。
Articles controlled by national laws and regulations (non-coordinated articles).
UKCA标志使用一般规则如下:
UKCA标志只能作为制造商或其授权代表(在相关法律允许的情况下)加贴在产品上;
在加贴UKCA标志时,制造商对产品符合相关法规要求承担全部责任;
制造商必须仅使用UKCA标志来证明产品符合相关的英国法规;
制造商不得将可能误解UKCA标志的含义或形式的任何标志放在第三方上;
制造商不得在产品上附加影响UKCA标志的可见性,易读性或含义的其他标志;
UKCA标志不能放在产品上,除非有在立法中作出特定需求。
The UKCA logo USES the following general rules:
The UKCA mark may only be affixed to the product as a manufacturer or its authorized representative (where permitted by applicable law);
When the UKCA logo is affixed, the manufacturer is fully responsible for compliance with the relevant regulatory requirements;
Manufacturers must use only the UKCA mark to demonstrate compliance with the relevant UK regulations;
The manufacturer shall not place any marks on third parties which may misunderstand the meaning or form of the UKCA mark;
The manufacturer shall not attach to the product other marks that affect the visibility, readability or meaning of the UKCA mark;
The UKCA logo cannot be placed on a product unless a specific requirement is made in legislation.
加贴UKCA标志图像的规则
Rules for attaching UKCA logo images
1. UKCA标志必须按规定的比例缩小或放大;
2. UKCA标志的高度至少为5mm,除非另有相关法规规定;
4. UKCA标志必须容易识别、清晰可见、耐久性高。
1. The UKCA mark must be reduced or enlarged to a specified scale;
2. The height of the UKCA mark shall be at least 5mm, unless otherwise specified by relevant regulations;
3. The UKCA logo must be easy to recognize, clearly visible and of high durability.
技术文档
Technical documentation
制造商或其授权代表必须保留技术文档证明其产品符合法规要求。市场监督或执法机构可随时要求制造商提供此信息。最长可在产品投放市场后10年内提供。
Manufacturers or their authorized representatives must maintain technical documentation to prove that their products comply with regulatory requirements. Market surveillance or law enforcement agencies may request this information from the manufacturer at any time.
It can be supplied up to 10 years after the product is put on the market.
记录保存要求将根据产品适用的具体法规而有所不同,一般需保留:
Record keeping requirements will vary according to the specific regulations applicable to the product and will generally be maintained as follows:
1. 产品的设计和制造记录;
2. 如何证明产品符合相关要求;
3. 制造商和任何存储设施的地址。
信息应以技术文件的形式存储,该文件可提供给市场监督机构。
1. Product design and manufacturing records;
2. How to prove that the product meets the relevant requirements;
3. Address of manufacturer and any storage facilities.
The information shall be stored in a technical document which may be made available to the market watchdog.
英国符合性声明
英国符合性声明是一份为合法使用UKCA标志的大多数产品制定的文件。在该文件中,制造商或其授权代表应:
1. 声明产品符合适用于特定产品的相关法规的要求;
2. 确保文件中包含制造商或其授权代表的名称和地址,以及有关产品和合格评定机构的信息。
3.制造商必须应要求向市场监督机构提供英国符合性声明。
4.在使用CE认证标志时,制造商还需要准备一份欧盟符合性声明。
UK declaration of conformity
The UK Statement of Compliance is a document developed for most products that legally use the UKCA logo.
In that document, the manufacturer or its authorized representative shall:
1. Declare that the product complies with the requirements of the relevant regulations applicable to the specific product;
2. Ensure that documents contain the name and address of the manufacturer or its authorized representative, as well as information about the product and conformity assessment body.
3. The manufacturer must provide the market watchdog with a STATEMENT of UK compliance upon request.
4. When using the CE mark, the manufacturer shall also prepare a declaration of COMPLIANCE with the EU.
我产品在英国已经销售了拥有CE证书,之后该怎么操作?
My product has been sold in The UK and I have the CE certificate. How should I operate after that?
你需要选择一条符合你组织需要的道路,你需要从这两个选项中选择一个;
You will need to choose the path which meets your organization’s needs and you will need to choose one of these two options;
- 继续与英国的认证机构合作,并立即申请UKCA标记。在这种情况下,您的CE标志将不再有效,您将无法将您的产品投放到欧盟27个成员国的市场上。一旦你有了UKCA,你就可以把你的产品投放到英国市场。
- Continue to work with your UK-based notified body and apply for UKCA marking immediately. In this case your CE marking will no longer be valid and you will be unable to place your products on the market in any of the EU 27 member states. Once you have the UKCA you will be able to place your products on the market in the UK.
- 选择与在欧盟27国注册的公告机构合作,以获得CE标志。这一点在英国脱欧后的有限时间内将得到欧盟和英国的承认。
- Chose to work with a notified body registered in an EU 27 member state to gain the CE marking. This will be recognized in the EU and also in the UK for a limited time after Brexit.
但是,如果您的产品需要公告机构提供第三方合格评定服务,并且该服务是由英国的认证机构执行的,则在“不交易”的情况下,您的CE标志将不再有效。这是因为英国的认证机构将不再得到欧盟的认可。
However, if your product requires a third party conformity assessment service to be provided by a notified body and the service is performed by a UK certification body, your CE mark will no longer be valid in the case of a "no trade".This is because UK certification bodies will no longer be recognised by the EU.
英国脱欧后实行UKCA认证重要信息:
The IMPLEMENTATION of UKCA certification after Brexit
(1)从欧盟27国第三方指定机构或者从英国第三方指定机构转移出认证,都必须在英国脱欧过渡期结束之前实施完成。
(2)欧盟27国第三方指定机构所实施的测试可以提交给英国认可机构,以便用于颁发UKCA认证。
(3)根据英国政府目前的指导方针,如果您的产品适用以下其中任何一项,那么,您的产品仍然可以在一个有限期限内在英国市场使用CE标志,这个期限暂时还没有确定,不过英国政府将会在这个期限终止之前给出正通知。
(4)关于测试要求的详细信息尚未列明,但是,预计测试要求将会反映或非常类似于现行的CE标志测试要求。可以同时进行CE标志测试和UKCA标志测试(EN标准),EN标准将适用于UKCA标志。
(5)由于英国在2020年12月31日正式退出欧盟之前,英国尚未颁布法律以实施UKCA标志,因此,在2021年1月1日之前,不能在产品上使用或放置UKCA标志。从2021年1月1日起,英国市场将在一个有限时期内认可带有CE标志的产品。一旦英国脱离欧盟,制造商们就会被告知他们有义务在产品上加注UKCA标志,并且,CE标志在有限认可期内将得到认可。
(6)同一款产品可显示多个符合性标志(包括CE和UKCA),以便进入全球市场。同一款产品标注多个符合性标志以适用不同的市场,这种情景比较常见。
(7)像CE标志一样,一旦英国颁布法律实行UKCA标志,就需要遵守相关详细规则,在产品标签上、包装上以及在说明手册中添加符合性标志。
(8)英国是公认的IECEE/IECEx CB报告及认证颁发国/认可国,因此,CB认证不受英国脱欧的影响。
(9)截至2016年,所有符合低电压指令(LVD)的产品均为自行声明产品。英国已经采纳该指令,并且该指令将适用于英国市场。唯一的区别是,将采用适用于英国市场准入的UKCA标志,而非采用适用于欧洲市场的CE标志。英国于2021年1月1日正式退出欧盟之后,将在一段有限期限内认可CE标志。
(10)已经具有CE认证的LVD、EMC、RoHS、WEEE测试报告,无需重新进行测试,也无需进行文件评审。对于已经自行出具声明的产品,制造商需在标注UKCA标志之前,起草一份符合性声明。
(11)同一份技术文件可同时用于获得UKCA标
The IMPLEMENTATION of UKCA certification after Brexit
(1) The transfer of certification from a third-party designated institution of the 27 EU countries or from a third-party designated institution of the UK must be completed before the end of the Brexit transition period.
(2) Tests carried out by third-party designations in eu27 may be submitted to UK accreditation bodies for UKCA certification.
(3) In accordance with the UK Government's current guidelines, if your product applies to any of the following, then your product may still use the CE mark in the UK market for a limited period, which has not yet been determined, but the UK Government will give positive notice of the termination of this period.
(4) Details of the test requirements have not been specified, but it is expected that the test requirements will reflect or be very similar to the current CE mark test requirements.
Both the CE mark test and the UKCA mark test (EN standard) can be performed, and the EN standard will apply to the UKCA mark.
(5) As the UK has not enacted legislation to implement the UKCA logo before its formal withdrawal from the EU on 31 December 2020, the UKCA logo cannot be used or placed on products before 1 January 2021.
Products bearing the CE mark will be recognised in the UK market for a limited period from 1 January 2021.
Once the UK leaves the EU, manufacturers will be told they have an obligation to put the UKCA mark on their products and that the CE mark will be recognised for a limited period.
(6) The same product can display more than one conformity mark (including CE and UKCA), so as to enter the global market.
It is common for the same product to be labeled with multiple compliance marks for different markets.
(7) Just like THE CE mark, once the UKCA mark is enacted and implemented in the UK, relevant detailed rules need to be observed to add the mark of conformity on the product label, packaging and in the instruction manual.
(8) The United Kingdom is a recognized IECEE/IECEx CB reporting and certification country/accreditation country. Therefore, CB certification is not affected by Brexit.
(9) As of 2016, all products conforming to low voltage instruction (LVD) are self-declared products.
The UK has adopted the directive and the directive will apply to the UK market.
The only difference is that the UKCA mark for UK market access will be used instead of the CE mark for the European market.
The UK will recognise the CE mark for a limited period of time after it formally leaves the EU on 1 January 2021.
(10) Test reports of LVD, EMC, RoHS and WEEE that have been CE certified do not need to be re-tested or reviewed.
For products that have already made their own declaration, the manufacturer will need to draw up a declaration of compliance before marking the UKCA logo.
(11) The same technical document may be used simultaneously to obtain the UKCA bid